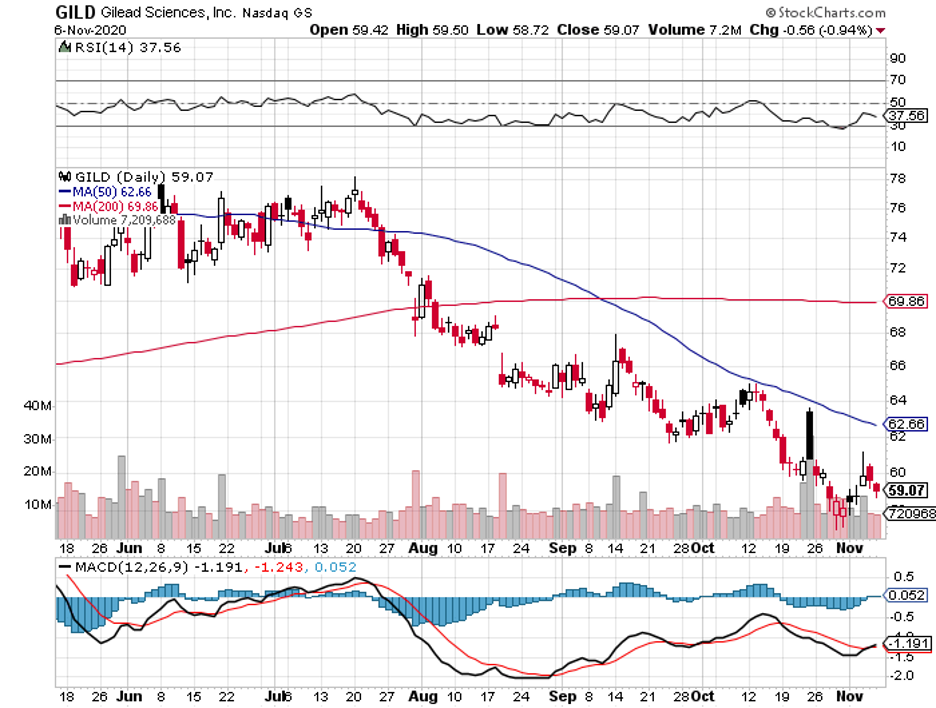
The fight against the coronavirus reached a major milestone when the US Food and Drug Administration (FDA) approved the first ever treatment for this deadly disease.
Unsurprisingly, the chosen leader for COVID-19 treatment to cross the full approval finish line is the same company that has been supplying the medication since the pandemic started: Gilead Sciences (GILD).
While Gilead’s Remdesivir has been widely used since January to treat severe cases of COVID-19, this FDA approval makes it official—a welcome piece of good news that pushed the stock up by 4% upon announcement.
Now, Gilead can broadly market Remdesivir under its official drug name, Veklury, to doctors and patients.
That means that other than the elderly and severe cases, Veklury can be marketed to COVID-19 patients as young as 12 years old.
Since Veklury gained approval, the drug has generated roughly $873 million in revenues.
Despite its limited market, this COVID-19 treatment actually ranked as Gilead’s second highest-selling drug in the third quarter of 2020—only behind the blockbuster HIV medication Biktarvy, which rose by 8% to contribute $4.55 billion.
As expected, Veklury’s popularity boosted Gilead’s 2020 performance.
Gilead’s total sales for the third quarter alone reached $6.5 billion, with $873 million coming from its brand new just-approved COVID-19 treatment Veklury.
For context, the company’s sales for the third quarter was only projected to grow by 2%. Veklury sales boosted this number to generate an 18% jump in revenue instead.
Clearly, Veklury injects a ray of hope in the declining sales for some of previous Gilead’s money makers like its hepatitis lineup, which saw a $210 million slide in revenue this quarter.
With this approval, Gilead is expected to pocket billions in Veklury sales as the company announced its plan to ramp up production to meet the global demand.
After all, governments are expected to stockpile the drug to be ready for future outbreaks.
In terms of its sustainability, Gilead is estimated to enjoy Veklury’s lucrative profits for a year or two until a COVID-19 vaccine gets fully approved or when herd immunity eventually kicks in.
Apart from Gilead, there are also other companies looking to cash in on this demand.
One of them is Regeneron Pharmaceuticals (REGN), which gained popularity after being used to fast track the COVID-19 recovery of Donald Trump during the campaign period. Another is Eli Lilly, which also applied for an emergency authorization for its antibody cocktail.
Most importantly, Veklury sets a promising precedent for other COVID-19 programs, particularly the vaccines.
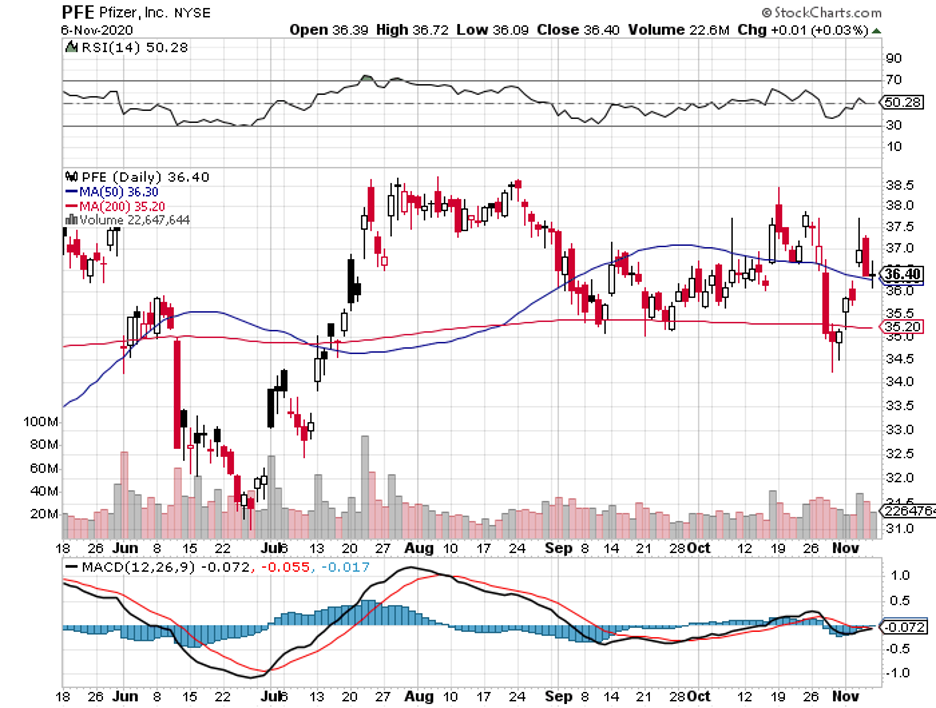
If the ongoing trials yield positive results, then the vaccines of Pfizer (PFE), AstraZeneca (AZN), Moderna (MRNA), and BioNTech (BNTX) could quickly receive emergency authorizations.
Meanwhile, Veklury is not the only pandemic-defying achievement of Gilead this year.
Even before the pandemic broke, Gilead’s strategy has consistently centered on acquisitions.
This plan was kickstarted with its $12 billion acquisition of Kite Pharma in 2017.
This investment has been paying off as the company continues growth in Asia, specifically in China.
By 2022, Gilead is projected to generate over $1 billion in sales from its Hepatitis B lineup in this region alone.
While 2020 has not been the best year for mergers and even acquisitions particularly in the biopharmaceutical sector, Gilead seems to not be letting the pandemic ruin its plans.
In March, Gilead completed its $4.9 billion acquisition of Forty-Seven in an effort to own the rights to a blockbuster cancer drug called Magrolimab. This product is anticipated to bring more than $3 billion in annual sales.
Recently, the company announced yet another massive $21 billion deal to acquire Immunomedics (IMMU)—a value that is nearly 30% of Gilead’s $70.4 billion market capitalization.
Gilead’s deal with Immunomedics adds another potential blockbuster drug in its oncology lineup: Trodelvy.
Once approved, Trodelvy is expected to rake in $4 billion annually—a profit that would eventually pay off the $21 billion that Gilead shelled out to acquire Immunomedics.
Looking at profits from its recent acquisitions, Gilead can rake in roughly $2 billion in quarterly revenue just for Trodelvy and Magrolimab alone.
Overall, Gilead’s product lineup has clearly shown significant growth.
Its core portfolio has been consistently strong, and the full FDA approval of Remdesivir offered the company a short-term boost.
In terms of long-term growth, Gilead maintains the capacity to provide significant cash flow for its shareholders.