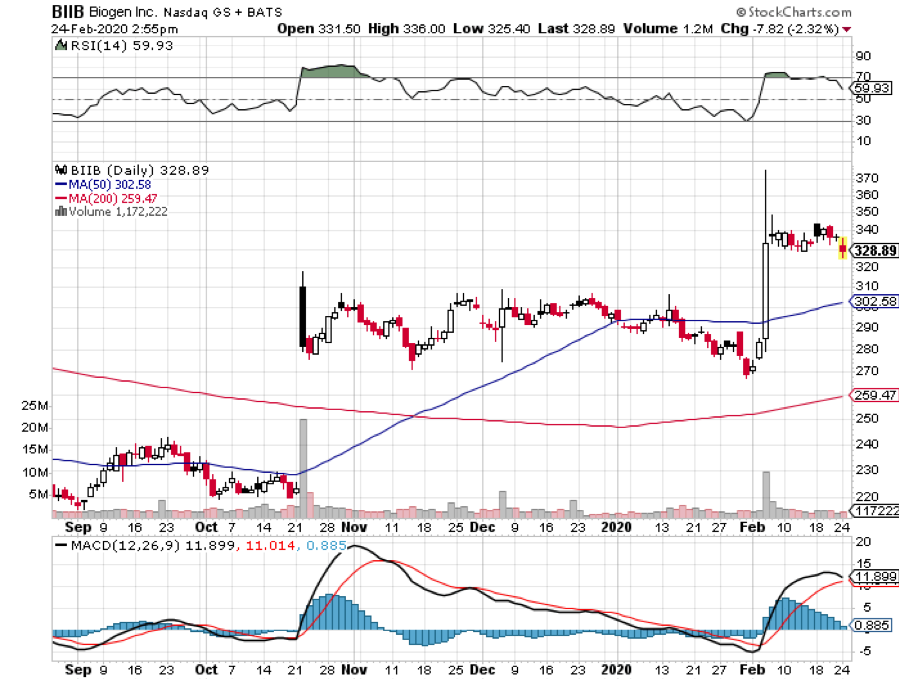
Coca-Cola (KO). Apple (APPL). American-Express (AXP). And now…. Biogen (BIIB).
Has the Oracle of Omaha turned into the Gambler of Omaha? Warren Buffett turned heads when news broke that Berkshire Hathaway bought 648,447 Biogen shares with a combined worth of $192.4 million.
With a huge question mark hanging over the biotechnology company for months now, this is an unusually risky move for an investment powerhouse known for its rock-solid strategies.
By Buffett standards though, $192.4 million is nothing but a drop in the over half-trillion-dollar bucket. This indicates that the conglomerate isn’t exactly betting the farm on the stock especially with questions on its Alzheimer’s cure still remaining unanswered.
Nonetheless, I can think of at least three reasons why Biogen stock attracted the fourth wealthiest man in the world.
A quick look at Biogen’s profile and the remarkable history of its blockbuster spinal muscular atrophy drug Spinraza immediately outshines the rest of the drugs in its portfolio.
Despite worries that Novartis’ (NOVN) newly released gene therapy Zolgensma would bump off Spinraza, the Biogen drug has been holding down its own, showing off a 16% jump year over year to hit $543 million in sales in the fourth quarter of 2019.
Another exciting development for the biotechnology company is its strengthened multiple sclerosis (MS) franchise.
While Biogen’s MS sales have been struggling to hit its usual stellar numbers recently, the company scored a huge victory earlier this month when the Patent Trial and Appeal Board of the US Patent and Trademark’s Office rejected Mylan’s (MYL) challenge against Biogen’s key patent for Tecfidera.
Since Tecfidera is one of Biogen’s top grossing treatments, this recent ruling secured that the MS bestseller won’t have to worry about generic competition until 2028. Apart from that, this win means that the company could count on a sustained earnings from this bestseller.
The third reason is arguably the most controversial, but possibly the most important one to date.
Following a near-death experience with its Alzheimer’s disease treatment Aducanumab last year, Biogen stunned the biotechnology world when it decided to resurrect the experiment based on new clinical data.
Given that Phase 3 is dubbed as the “graveyard” stage since practically 99% of experimental drugs fail here, Biogen’s move to keep pushing despite its expensive failures has investors baffled and excited at the same time.
Basically, the problem in finding an Alzheimer’s cure is our inability to fully grasp the underlying science.
What we know so far is that the disease is caused by plaques in the brain. Now, experts normally pursue two leads. They either go after the plaques and search for ways to cure those or take a step back and look at the possible causes of the plaques and target those instead.
Biogen worked on these hypotheses up until March 2019, when they announced that the trial had been discontinued.
However, the company went back months after and disclosed that they’ll resume the study. After combing through their data, they noticed that the group of patients in the earlier stage of the disease actually responded well on Aducanumab.
One notable effect of the experimental Alzheimer’s disease medication is that it slowed down the patients’ cognitive decline.
While it’s certainly reasonable to question these retrospective ad-hoc reports especially since companies tend to mine for data just to recoup their investments on the trials, Biogen’s ability to back their claims with verifiable numbers makes them more credible.
This gamble is far from a slam dunk but an FDA approval for Aducanumab would guarantee the addition of another blockbuster drug in the Biogen lineup.
And though the company has a long way to go, this Alzheimer’s gamble is definitely a high-risk, high-reward opportunity, with an estimated $10 billion in peak sales.
Aside from Aducanumab, Biogen has other promising candidates. The notable ones include rare amyotrophic lateral sclerosis treatment BIIB067 and ischemic stroke medication BIIB093.
Buffett has never been much of a gambler when it comes to his career. With this recent investment on Biogen stock though, the billionaire investor appears to have given his stamp of approval to what can only be the riskiest bets in Berkshire’s history.