Two major issues are in the root of the panic caused by every new infectious disease.
The first is because of the uncontrollable spread of the virus, affecting thousands of people in a short span of time. This consequently pushes the public to question the capacity of their government to provide the proper healthcare to every patient, causing further confusion, alarm, and eventually, economic upheaval.
The second is the overpowering uncertainty that looms over not only the people directly affected by the disease but also the rest of the world as we feel like sitting ducks, wondering who the virus will infect next.
Both effects have become apparent with the news of a new coronavirus, known as 2019-nCoV, which originated in Wuhan, China.
The public’s reaction has come so close to mass hysteria, especially since the virus has already taken over 400 lives and infected more than 20,000 people worldwide.
The speed by which the disease is spreading is also alarming. The 2019-nCoV getting transmitted from one person to another almost as fast as the highly transmissible flu, which was not the case for its slower-moving cousins Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS).
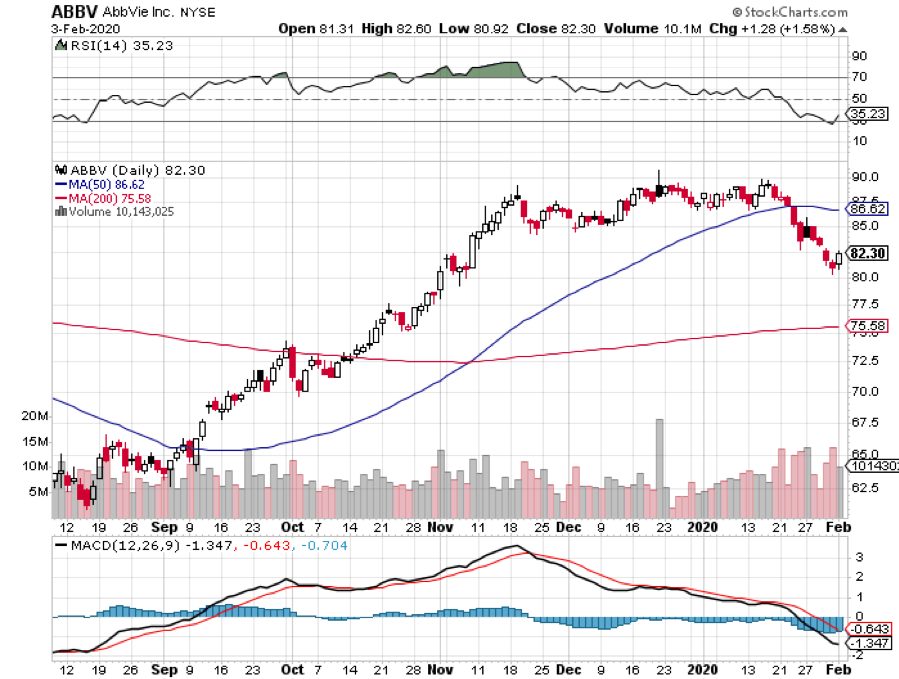
In response to this alarming situation, the Chinese government along with the medical community has turned to unconventional means to find a cure — and giant biotechnology company AbbVie (ABBV) is at the forefront of these efforts today.
One measure taken by the health experts is using AbbVie’s off-label HIV drug, called Kaletra (aka Aluvia), to treat 2019-nCoV patients.
Kaletra has two components valuable in battling the coronavirus: lopinavir and ritonavir. Both have the capacity to block HIV viral replication.
The twice-a-day Kaletra regimen comprises taking the HIV drug and undergoing infusions of interferon, a protein that triggers the immune system.
Although the AbbVie drug has yet to be officially declared as an approved cure for the 2019-nCoV, more and more health experts in China are already using it.
The belief on this treatment’s efficacy stemmed from the statement of the head at Peking University First Hospital, who shared that he contracted 2019-nCoV but cured himself by taking Kaletra.
This Chinese doctor is currently part of the national team of experts sent by the Chinese government to tend to those in Wuhan. Aside from him, Shanghai authorities disclosed that they have already adopted the HIV treatment in handling their own patients.
However, this isn’t the first time that Kaletra was deployed as a treatment against a coronavirus.
In 2004, Kaletra was used to cure the patients of SARS-CoV. This is promising since SARS, which is also caused by a coronavirus, bears a close resemblance to the 2019-nCoV. Experts tested the drug to help treat MERS patients as well.
A roadblock for the Kaletra cure is the scarcity of the drug in China.
A lot of health professionals disclosed that they have been struggling to get their hands on the AbbVie off-patent product. In response, AbbVie donated approximately $1.5 million worth of Kaletra to help contain the 2019-nCoV.
Other biotechnology behemoths have followed AbbVie’s lead of donating funds to help with the research in China. The list includes Roche Holding (RHHBY) and Gilead Sciences (GILD).
Meanwhile, this piece of promising news is not lost in the market. In fact, AbbVie shares pushed higher the moment Kaletra’s efficacy against the 2019-nCoV was revealed.
Apart from that, this development has reminded investors of AbbVie’s promising growth lately.
Unfortunately, most of the company’s growth came from acquisitions — a path where AbbVie has an abysmal track record. More often than not, investors fear that the company overpays in deals that fail to reap the promised rewards.
Case in point: AbbVie’s hefty $63 billion acquisition of Botox maker Allergan, which is due to be finalized early 2020.
In moving to acquire a company that only has one consistent blockbuster product, experts believe that AbbVie might have pulled the trigger too soon without considering other options.
While this high-profile agreement has been such a hot topic in the investing community, I think it’s a good move towards diversifying AbbVie’s sales and injecting additional growth.
Remember, AbbVie is in desperate need for a new outlet in light of the dwindling Humira sales. Adding a high-selling and established industry leader like Botox to the mix would make AbbVie more diversified than ever, making it safer.
More importantly, AbbVie ensured that the $63 billion acquisition doesn’t run the company to the ground — and its precautionary measures showed.
The company has been consistent in its net income, reporting over $5 billion annually in the past three years. It also raked in $12.8 billion in free cash flow in 2018, proving that AbbVie is well-positioned to acquire more companies and be more creative in looking for growth targets.
Hence, I view this biotechnology stock as an underrated buy that would appeal to bargain hunters willing to hang onto it for the years to come.